Hydrogen embrittlement Chapter 2
13350 Views |
In the previous chapter, I have written about Hydrogen Embrittlement and delayed fracture. This chapter will be about why material became brittle when Hydrogen entered the steel.
The mechanism of Hydrogen Embrittlement still be a mystery
I am also wondering whether I should write this article or not, there are theories of mechanism behind the state when steel became brittle when Hydrogen entered. Hydrogen is a very small element in our world where we can find it everywhere even the air, compare to atomic size of metal which is Fe(Iron). It can move freely in within it, making it difficult to detect hydrogen.
There are many factors that cause Hydrogen Embrittlement including time, temperature, stress, material strength and etc. Made this issue became very complicated.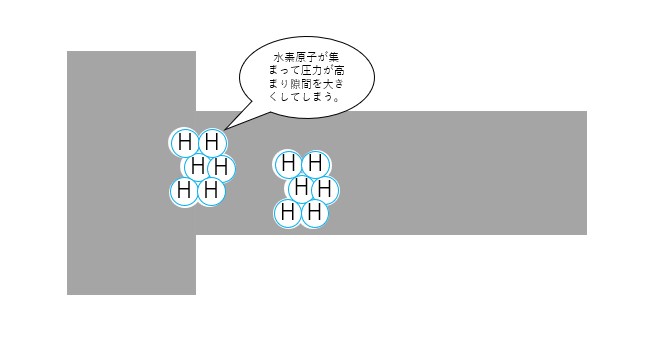
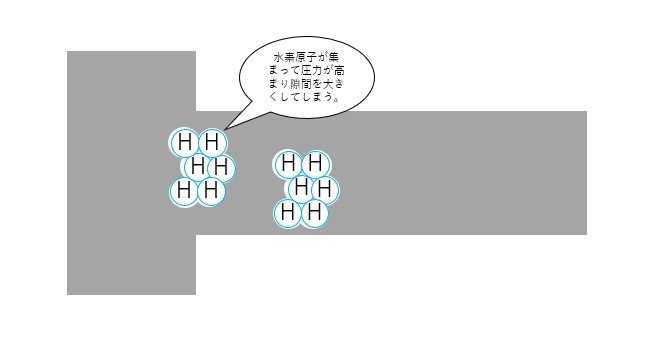
Theory ① High pressure hydrogen gas theory
Hydrogen atoms gather in tiny gaps in the steel and turn into hydrogen molecules, applying pressure to the gaps and making them larger.
Theory ②: Decreased bonding strength between atoms
Hydrogen atoms enter the iron atoms that trying to stick together to maintain their tensile strength, hydrogen will reduce the bonding strength between the iron atoms and causing them to crack and break easily.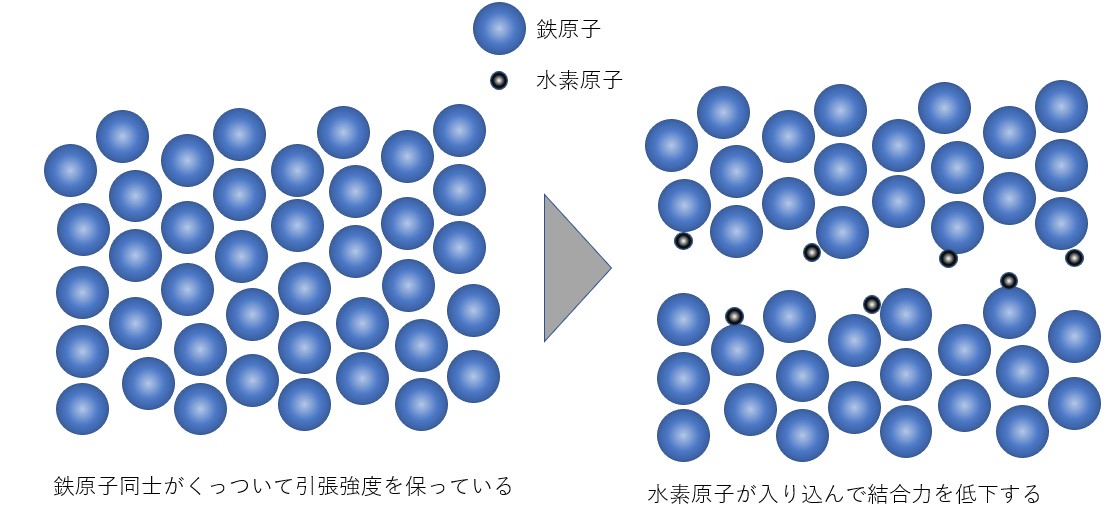
There are other theories as well, such as the hydrogen adsorption theory, but these have not been completely proved for the reasons mentioned above.
However, it seems certain that hydrogen makes the steel brittle, so if the hydrogen is removed through baking, the strength of the steel will be maintained.
Summary
Delayed fracture caused by hydrogen embrittlement is a very frightening phenomenon for manufacturing. However, if you are plating high-strength bolts, there is no problem as long as the strength classification is up to 10.9 and baking treatment is performed afterwards.
If you really want to apply rust prevention treatment to something with a strength of 12.9 or higher, please feel free to contact us as there are treatment methods that do not cause hydrogen embrittlement, such as Geomet®.



